
Personal Chair
- About
-
- Email Address
- h.deng@abdn.ac.uk
- Telephone Number
- +44 (0)1224 272953
- Office Address
- School/Department
- School of Natural and Computing Sciences
Biography
08/2022 - Personal Chair, University of Aberdeen, UK
08/2018 - 07/2022 Reader, University of Aberdeen, UK
08/2014 - 07/2018 Senior Lecturer, University of Aberdeen, UK
10/2008 - 07/2014 Lecturer, University of Aberdeen, UK
06/2002 - 09/2008 Postdoctoral fellow, University of St Andrews, UK
1999 - 2002 Ph.D. University of Wales Swansea, UK
Memberships and Affiliations
- Internal Memberships
-
Director of Research
PGR coordinator
CM2514 course coordinator
- External Memberships
-
BBSRC pool of expert
MRC and BBSRC panel members
UKRI FLF peer review colleague
External reviewer for a series of peer-reviewed journals within my area of interest, including Nature, Nat. Chen. ACIE, Sci. Adv. Chem. Sci., Chem Comm., ACS Chem. Biol., ChemBioChem, Marine Biotechnology, Org & Biomol. Chem, FEMS Microbiology Ecology, Chemistry today, Marine Drugs, Fish and Shellfish Immunology, and Virulence.
A reviewer of research councils, BBSRC (UK) and FCT (Fundação para a Ciência e a Tecnologia in the field of Chemistry and Biochemistry, 2010-2011), Fonds de recherche du Québec - Nature et technologies, Québec, Canada (2015-2016) and charities, Leverhulme Trust (UK).
- Research
-
Research Overview
My research carries on the great tradition of UK natural product biosynthesis activity. We have all the required skills to succeed in such an enterprise, involving discovery of new families of specialised metabolites, bacterial genomics, chemical synthesis and the associated molecular biology to manipulate genomes to enzymology and the reconstruction of biochemistry. All of this is focussed on understanding the molecular basis and mechanism of natural products assembly.
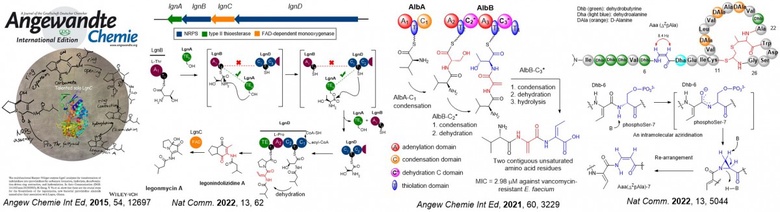
Recent contributions involve the understanding of a group of unsaturated amino acids called dehydroamino acids (dhAAs). dhAAs are key components in many peptidyl therapeutics and versatile building blocks in peptide chemistry where organic chemists generate peptidyl derivatives through Michael additions, cross couplings and cycloaddition. They are used as bio-orthogonal handles for later stage modification of biomolecules. However, accessing dhAAs in structurally complex molecules presents a synthetic challenge, often resulting in poor atomic economy.
Among approximately 40 dhAAs found in the natural product inventory, only two, dehydroalanine (Dha) and (Z)-dehydrobutyrine (ZDhb), are well-studied. In this context our lab has elucidated the formation of several understudied dhAAs in newly discovered bacterial peptide-related metabolites, such as pyrrolizidine alkaloids (PAs) (Angew Chem. Int. Ed. 2015, 54, 12697, Nat. Comm. 2022, 13, 62), short dehydrated non-ribosomal peptides (Angew Chemie. Int. Ed. 2021, 60, 3229) and a new family of ribosomally synthesized and posttranslationally modified peptides (RiPPs) (Nat. Comm. 2022, 13, 5044) as shown below. Apart from this, we have also accumulated an outstanding body of work from bioactive natural product discovery to new enzymology as evidenced in our publication profile.
The discovery we made offers an alternative to rational engineering of pathways to generate bioactive peptides. As such we have obtained grants from various funding bodies (i.e. UKRI, Leverhulm Trust, Royal Society, Royal Society of Edinburgh, IBIOIC, EC) to further investigate potential applications of novel enzymes identified in these pathways towards the development of pharmaceuticals.
Research Areas

Biological and Environmental Sciences

Chemistry
Research Specialisms
- Industrial Biotechnology
- Biochemistry
- Organic Chemistry
- Applied Chemistry
Our research specialisms are based on the Higher Education Classification of Subjects (HECoS) which is HESA open data, published under the Creative Commons Attribution 4.0 International licence.
Past Research
My team has accumulated a good body of work on bioactive natural product discovery (from polyketides and peptides to alkaloids) and their biosynthesis, and new enzyme investigation as shown below. Collectively it is a combination of both its range and illustration and these new organic molecules and new enzymes my team has discovered will find applications for the development of pharmaceuticals and agrochemicals or new ways to biosciences.

Collaborations
Professor Mathew Jenner, Department of Chemistry, University of Warwick, UK
Professor Steven Cobb, Department of Chemistry, University of Durham, UK
Dr Jioji Tabudravu, School of Natural Sciences, University of Central Lancashire, UK
Dr David Clarke, EastChem, School of Chemistry, University of Edinburgh, Edinburgh, EH9 3FJ, UK
Professor Bruce Milne, Department of Physics, University of Coimbra, Rua Larga, 3004-516, Coimbra, Portugal
Professor Yi Yu, School of Pharmaceutical Sciences, Wuhan University, Wuhan 430071, China
Professor Kwaku Kyeremeh, Department of Chemistry, University of Ghana, Ghana
NCIMB Ltd and Ingenza Ltd
Funding and Grants
We gratefully thank the funding bodies below for financial supports of our research
MRC, BBSRC, Leverhulm Trust, EC, IBIOIC, The Royal Society and The Royal Society of Edinburgh.

- Teaching
-
Teaching Responsibilities
CM4518 Biological origin of natural products
CM1020 Chemistry for Biosciences 1
CM1512 Chemistry for Biosciences 2
CM3534 Organic and Biological Chemistry
- Publications
-
Page 2 of 2 Results 51 to 100 of 100
Profiling of secondary metabolite gene clusters regulated by LaeA in Aspergillus niger FGSC A1279 based on genome sequencing and transcriptome analysis
Research in Microbiology, vol. 169, no. 2, pp. 67-77Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.1016/j.resmic.2017.10.002
(±)-Hippolide J – A Pair of Unusual Antifungal Enantiomeric Sesterterpenoids from the Marine Sponge Hippospongia lachne
European Journal of Organic Chemistry, vol. 2017, no. 24, pp. 3421-3426Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.1002/ejoc.201700248
- [ONLINE] View publication in Scopus
Targeted Dereplication of Microbial Natural Products by High-Resolution MS and Predicted LC Retention Time
Journal of Natural Products, vol. 80, no. 5, pp. 1370-1377Contributions to Journals: ArticlesDissection of the neocarazostatin: a C4 alkyl side chain biosynthesis by in vitro reconstitution
Organic & Biomolecular Chemistry, vol. 15, no. 18, pp. 3843-3848Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.1039/c7ob00617a
Pseudochelin A, a siderophore of Pseudoalteromonas piscicida S2040
Tetrahedron, vol. 73, no. 18, pp. 2633-2637Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.1016/j.tet.2017.03.051
- [OPEN ACCESS] http://aura.abdn.ac.uk/bitstream/2164/10213/1/Manuscript_reviewed_JTdocx.docx
- [ONLINE] View publication in Scopus
A ThDP-dependent enzymatic carboligation reaction involved in Neocarazostatin A tricyclic carbazole formation
Organic & Biomolecular Chemistry, vol. 14, no. 37, pp. 8679-8684Contributions to Journals: ArticlesBiological fluorination from the sea: discovery of a SAM-dependent nucleophilic fluorinating enzyme from the marine-derived bacterium Streptomyces xinghaiensis NRRL B24674
RSC Advances, vol. 32, no. 6, pp. 27047-27051Contributions to Journals: ArticlesBiosynthesis of Neocarazostatin A Reveals the Sequential Carbazole Prenylation and Hydroxylation in the Tailoring Steps
Chemistry & Biology, vol. 22, no. 12, pp. 1633-1642Contributions to Journals: ArticlesDiscovery of a Single Monooxygenase that Catalyzes Carbamate Formation and Ring Contraction in the Biosynthesis of the Legonmycins
Angewandte Chemie International Edition, vol. 54, no. 43, pp. 12697-12701Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.1002/anie.201502902
Chaxapeptin, a Lasso Peptide from Extremotolerant Streptomyces leeuwenhoekii Strain C58 from the Hyperarid Atacama Desert
Journal of Organic Chemistry, vol. 80, no. 20, pp. 10252-10260Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.1021/acs.joc.5b01878
Identification and characterization of the biosynthetic gene cluster of thiolutin, a tumor angiogenesis inhibitor, in Saccharothrix algeriensis NRRL B-24137
Anti-Cancer Agents in Medicinal Chemistry, vol. 15, no. 3, pp. 277-284Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.2174/1871520614666141027145200
Enzymatic fluorination and biotechnological development of the fluorinase
Chemical Reviews, vol. 115, no. 2, pp. 634-649Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.1021/cr500209t
Diversity of organic structures of marine microbial origin with drug potential
Phycotoxins: Chemistry and Biochemistry. Botana, L., Alfonso, A. (eds.). 2 edition. Blackwell Science, pp. 361-380, 20 pagesChapters in Books, Reports and Conference Proceedings: ChaptersDraft Genome Sequence of Streptomyces sp. Strain CT34, Isolated from a Ghanaian Soil Sample
Genome Announcements, vol. 3, no. 1, 01508-14Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.1128/genomeA.01508-14
- [OPEN ACCESS] http://aura.abdn.ac.uk/bitstream/2164/4432/1/e01508_14.full.pdf
Editorial, thematic issue: discovery and biosynthesis of natural products with anti-cancer activities
Anti-Cancer Agents in Medicinal Chemistry, vol. 15, no. 3, pp. 276Contributions to Journals: Editorials- [ONLINE] DOI: https://doi.org/10.2174/187152061503150313231442
Identification of a fluorometabolite from Streptomyces sp. MA37: (2R,3S,4S)-5-Fluoro-2,3,4-trihydroxypentanoic acid
Chemical Science, vol. 6, no. 2, pp. 1414-1419Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.1039/C4SC03540B
- [OPEN ACCESS] http://aura.abdn.ac.uk/bitstream/2164/4436/1/c4sc03540b.pdf
Legonaridin, a new member of linaridin RiPP from a Ghanaian Streptomyces Isolate
Organic & Biomolecular Chemistry, vol. 13, no. 37, pp. 9585-9592Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.1039/C5OB01269D
- [OPEN ACCESS] http://aura.abdn.ac.uk/bitstream/2164/6288/1/Legonaridin_manuscript_OBC.pdf
Butrepyrazinone, a New Pyrazinone with an Unusual Methylation Pattern from a Ghanaian Verrucosispora sp. K51G
Marine Drugs, vol. 12, no. 10, pp. 5197-5208Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.3390/md12105197
- [OPEN ACCESS] http://aura.abdn.ac.uk/bitstream/2164/3737/1/marinedrugs_12_05197.pdf
Fluoroacetate biosynthesis from the marine-derived bacterium Streptomyces xinghaiensis NRRL B-24674
Organic & Biomolecular Chemistry, vol. 27, no. 12, pp. 4828-4831Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.1039/c4ob00970c
Disruption of a methyltransferase gene in actinomycin G gene cluster in Streptomyces iakyrus increases the production of phenazinomycin
FEMS Microbiology Letters, vol. 352, no. 1, pp. 62-68Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.1111/1574-6968.12370
Butremycin, the 3-Hydroxyl Derivative of Ikarugamycin and a Protonated Aromatic Tautomer of 5′-Methylthioinosine from a Ghanaian Micromonospora sp. K310
Marine Drugs, vol. 12, no. 2, pp. 999-1012Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.3390/md12020999
- [OPEN ACCESS] http://aura.abdn.ac.uk/bitstream/2164/3125/1/marinedrugs_12_00999.pdf
Identification of Fluorinases from Streptomyces sp MA37, Norcardia brasiliensis, and Actinoplanes sp N902-109 by Genome Mining
ChemBioChem, vol. 15, no. 3, pp. 364-368Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.1002/cbic.201300732
Action in pairs: Two tandem genes in the fish pathogen Yersinia ruckeri are virulence factors
Virulence, vol. 5, no. 5, pp. 585-586Contributions to Journals: Editorials- [ONLINE] DOI: https://doi.org/10.4161/viru.29787
Dithiolopyrrolone Natural Products: Isolation, Synthesis and Biosynthesis
Marine Drugs, vol. 11, no. 10, pp. 3970-3997Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.3390/md11103970
- [OPEN ACCESS] http://aura.abdn.ac.uk/bitstream/2164/2972/1/marinedrugs_11_03970_1_.pdf
Tianchimycins A-B, 16-membered macrolides from the rare actinomycete Saccharothrix xinjiangensis
Tetrahedron, vol. 69, no. 30, pp. 6060-6064Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.1016/j.tet.2013.05.094
Identification and characterization of the actinomycin G gene cluster in Streptomyces iakyrus
Molecular BioSystems, vol. 9, no. 6, pp. 1286-1289Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.1039/c3mb70081j
The fish pathogen Yersinia ruckeri produces holomycin and uses an RNA methyltransferase for self-resistance
The Journal of Biological Chemistry, vol. 288, no. 21, pp. 14688-14697Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.1074/jbc.M112.448415
Mining complex bacteria media for all fluorinated compounds made possible by using HPLC Coupled Parallel to Fluorine-Specific and Molecular Specific Detection
Journal of Analytical Atomic Spectrometry, vol. 28, no. 6, pp. 877-882Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.1039/C3JA50086A
Fluorine speciation analysis using reverse phase liquid chromatography coupled off-line to continuum source molecular absorption spectrometry (CS-MAS): identification and quantification of novel fluorinated organic compounds in environmental and biological samples
Analytical Chemistry, vol. 84, no. 14, pp. 6213-6219Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.1021/ac301201y
Diverse Metabolic Profiles of a Streptomyces Strain Isolated from a Hyper-arid Environment
Journal of Natural Products, vol. 74, no. 9, pp. 1965-1971Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.1021/np200470u
Chaxamycins A-D, Bioactive Ansamycins from a Hyper-arid Desert Streptomyces sp
Journal of Natural Products, vol. 74, no. 6, pp. 1491-1499Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.1021/np200320u
Identification and heterologous expression of the biosynthetic gene cluster for holomycin produced by Streptomyces clavuligerus
Process Biochemistry, vol. 46, no. 3, pp. 811-816Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.1016/j.procbio.2010.11.024
An enzymatic route to 5-deoxy-5-[18F]fluoro-D-ribose, a [18F]-fluorinated sugar for PET imaging
Chemical Communications, vol. 46, no. 1, pp. 139-141Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.1039/b919364b
Mechanistic insights into water activation in SAM hydroxide adenosyltransferase (duf-62)
ChemBioChem, vol. 10, no. 15, pp. 2455-2459Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.1002/cbic.200900369
In vitro reconstituted biotransformation of 4-fluorothreonine from fluoride ion: Application of the fluorinase
Chemistry & Biology, vol. 15, no. 12, pp. 1268-1276Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.1016/j.chembiol.2008.10.012
The fluorinase, the chlorinase and the duf-62 enzymes
Current Opinion in Chemical Biology, vol. 12, no. 5, pp. 582-592Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.1016/j.cbpa.2008.06.036
S-Adenosyl-L-methionine:hydroxide adenosyltransferase: a SAM enzyme
Angewandte Chemie International Edition, vol. 47, no. 29, pp. 5357-5361Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.1002/anie.200800794
Fluorine and Health: Biological Fluorination in Streptomyces cattleya: The Fluorinase
Elsevier. 1st edition, pp. 761-777, 17 pagesChapters in Books, Reports and Conference Proceedings: Chapters- [ONLINE] DOI: https://doi.org/10.1016/B978-0-444-53086-8.00018-7
The identification of (3R, 4S)-5-fluoro-5-deoxy-D-ribulose-1-phosphate as an intermediate in fluorometabolite biosynthesis in Streptomyces cattleya
Bioorganic Chemistry, vol. 35, no. 5, pp. 375-385Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.1016/j.bioorg.2007.04.001
The fluorinase from Streptomyces cattleya is also a chlorinase
Angewandte Chemie International Edition, vol. 45, no. 5, pp. 759-762Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.1002/anie.200503582
Fluorinase mediated C--18 F bond formation, an enzymatic tool for PET labelling
Chemical Communications, no. 6, pp. 652-654Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.1039/B516861A
Substrate specificity in enzymatic fluorination. The fluorinase from Streptomyces cattleya accepts 2'-deoxyadenosine substrates
Organic & Biomolecular Chemistry, no. 8, pp. 1458-1460Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.1039/B600574H
The identification of 5'-fluoro-5-deoxyinosine as a shunt product in cell free extracts of Streptomyces cattleya
Bioorganic Chemistry, vol. 33, no. 5, pp. 393-401Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.1016/j.bioorg.2005.07.002
Enzymatic fluorination in Streptomyces cattleya takes place with an inversion of configuration consistent with an SN2 reaction mechanism
ChemBioChem, vol. 5, no. 5, pp. 685-690Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.1002/cbic.200300839
Crystal structure and mechanism of a bacterial fluorinating enzyme
Nature, vol. 427, no. 6974, pp. 561-565Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.1038/nature02280
Identification of 5-fluoro-5-deoxy-D-ribose-1-phosphate as an intermediate in fluorometabolite biosynthesis in Streptomyces cattleya
Chemical Communications, vol. 5, pp. 592-593Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.1039/b400754a
Fluorometabolite biosynthesis and the fluorinase from Streptomyces cattleya
Natural Product Reports, vol. 21, no. 6, pp. 773-784Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.1039/B415087M
The first enzymatic method for C–18F bond formation: the synthesis of 5′‐[18F]‐fluoro‐5′‐deoxyadenosine for imaging with PET
Journal of Labelled Compounds and Radiopharmaceuticals, vol. 46, no. 13, pp. 1181-1189Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.1002/jlcr.779
Isolation and characterisation of 5'-fluorodeoxyadenosine synthase, a fluorination enzyme from Streptomyces cattleya
FEBS LETTERS, vol. 547, no. 1-3, pp. 111-114Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.1016/s0014-5793(03)00688-4
Crystallization and X-ray diffraction of 5'-fluoro-5'-deoxyadenosine synthase, a fluorination enzyme from Streptomyces cattleya
Acta Crystallographica Section D: Biological Crystallography, vol. 59, no. 12, pp. 2292-2293Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.1107/S0907444903019826
