Patient and Public Involvement at the School of Medicine, Medical Sciences and Nutrition
University Commitment
The University of Aberdeen is equally committed to supporting and encouraging researchers to involve members of the public in research activities, as well as providing opportunities for members of the public to connect with and engage in our research.
What is Patient and Public Involvement in Research?
Patient and Public Involvement (also called PPI) is the term we use to describe working with members of the public and patients who will contribute to how research is designed, conducted and disseminated. It ensures that research stays accessible and relevant to public needs.
For all Patient and Public Involvement (PPI) enquiries, please contact Diane McCosh.
Upcoming events
Online Lunch & Learn sessions: These fortnightly online sessions, running from January to June 2025 via Microsoft Teams, aim to bring together anyone interested in being involved with research, whether you are a patient partner, a community member or a non-academic. Find out more about our upcoming Lunch & Learn sessions here.
PPI Resources for Researchers
- PPI in Research
-
What is Patient and Public Involvement in research?

Patient and public involvement in research differs from public engagement with research, in which researchers share and discuss their research with different audiences through forums like public events, festivals and social media or targeted community activities.
It is also different to research participation, in which researchers recruit and consent people to take part in research study activities, for example by completing a survey, providing clinical samples or taking part in an intervention trial. Unlike research participation, ethical approval is not needed for the majority of public involvement activities.
Why is Patient and Public Involvement in research so important?
We have long recognised that collaborating with patients, carers, community and charity organisations and individuals with relevant experience positively impacts on all stages of research. Our goal is to ensure that patient and public involvement in research leads to:
- Identification of the most relevant research priorities.
- Better design, management and undertaking of research projects.
- Improved interpretation of findings within given context.
- Wider dissemination and sharing of findings to public and key stakeholders.
- A change to clinical practice or policy by implementation of research findings.
- Continual evaluation of impact from research.
- Getting Started
-
Where to begin?
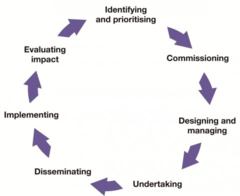
Whether you are just beginning to develop an idea for a research project or about to begin data analysis, you can involve members of the public at any point in the Research Cycle. It is never too early or too late to incorporate these activities into your research!
For example, you could:
- Host a priority setting exercise to determine what are the perceived research needs and gaps for your patient/public population.
- Develop a grant application with named members of an advisory group to determine design of project, key outcomes, planned activities and timeline.
- Involve patients or public in ethics and protocol development, participant recruitment and retention, and materials (e.g., leaflets, surveys, consent forms, website/social media material).
- Work with patients or public on creating the data analysis plan and interpreting findings from perspective of their lived experience.
- Disseminate and evaluate findings alongside patients or public to both academic and non-academic audiences.
Who can help?
If you have never undertaken PPI activities or if you have but want to further develop your skills, there are a number of resources available to you at the University of Aberdeen within the School of Medicine, Medical Sciences and Nutrition (SMMSN), including:
- A new SMMSN patient and public research interest group has been established and this group is available to researchers as a resource to help facilitate engagement with PPI. Please contact Diane McCosh if you would like further information.
- An internal fund is available to University of Aberdeen researchers to support a range of PPI activities. Applications are assessed competitively. More information can be found at the Patient and Public Involvement Fund .
- Regular training on effective and practical PPI and Public Engagement is provided through the Grants Academy ResearchBite series as well as the Public Engagement Research Unit (PERU) .
- Research and Innovation staff Juliette Snow and Despoina Kaloriti can advise researchers planning and developing grant applications on any funder-specific PPI requirements including eligible costs that can be requested to support PPI activities.
- A number of SMMSN colleagues have extensive PPI experience and are available for consultation. Please contact Diane McCosh if you would like more information.
- On occasion external funders launch ad hoc calls focussed on PPI activities; these opportunities are promoted through the Grants Academy and the PERU team can offer hands-on support to help prepare competitive proposals.
*Image of Research Cycle from NIHR - Involve
- External Resources & Workshops
-
A list of helpful external resources

- The National Institute for Health Research (NIHR) , the nation's largest funder of health and care research, providing the people, facilities and technology for research to thrive.
- The INVOLVE website offers a multitude of resources to assist in involving patients and public across all stages of the research cycle, including the National Standards for Public Involvement . Key pages include 'top tips' , as well as information on the payment and recognition for public involvement . This includes practical advice on budgeting for public involvement activities and calculating costs to include in grant applications. They also provide a comprehensive toolkit for writing a plain English , or “lay” summary in a clear and accessible language.
- Research charities like Cancer Research UK , Diabetes UK and Versus Arthritis provide useful information and guidance for researchers on patient and public involvement in research. In addition, NHS Grampian Research and Development is committed to improving public awareness and involvement in clinical research. They have a webpage dedicated to clinical research to support both researchers and the public, with news, events and current research projects.
- The James Lind Alliance “brings patients, carers and clinicians together in Priority Setting Partnerships to identify and prioritise the Top 10 unanswered questions” for a wide-range of health care conditions, from Acne to Womb Cancer. This is a wonderful resource, especially when at the beginning stages of idea or grant application development.
- SHARE is the Scottish Health Research Register - which was created by NHS Research Scotland to establish a register of people interested in participating in health research. SHARE can be accessed for PPI activities, although costs of using SHARE for PPI will apply and vary from project to project.
You may find the following useful if you are also interested in conducting Public Engagement activities.
National Co-ordinating Centre for Public Engagement
WORKSHOPS
ResearchBites Patient and Public Involvement Mini Series
This lunchtime mini-series will provide an exciting opportunity to hear from colleagues about their experiences working with patients and the public in research. The series will present an in-depth look into approaches used across the research cycle - from a pre-funded to funded stage. Colleagues, together with research partners, will share valuable lessons learned, as well as honestly speak about the benefits and challenges of undertaking work together.
Session 5: How PPI can enhance your Research:
Original date postponed, new date t.b.cThis first session with Dr Kathryn Martin and patient partners covers PAINSTORM: How Effective PPI Can Enhance Research Design and Funding Application. Sign up here .
If you have any queries, please do not hesitate to contact Gillian Cooper
- The National Institute for Health Research (NIHR) , the nation's largest funder of health and care research, providing the people, facilities and technology for research to thrive.
- For Members of the Public
-
Thank you for your interest in Patient and Public Involvement activities at the University of Aberdeen.
At this time, all we need is your full name and email address. You can enter these in the form below.
We will use these details to share more information with you and to invite you to Patient and Public Involvement events at the University in the future.
If you have more questions or would like more information, please email involve.grampian@abdn.ac.uk or contact Dr Kathryn Martin 01224 437117
Meet the PPI Partners
The School of Medicine, Medical Sciences & Nutrition has a diverse core Patient & Public Involvement group made up of twelve members who meet four times a year and regularly engage with researchers within the School/NHS to ensure the work is meaningful and impactful.
- Elizabeth Chrystall
-
I am 74 years old and live in Kintore with my partner of 30 years. Having worked in HR roles in the oil industry, public sector, wholesale distribution and transport companies, I became a self-employed HR Consultant for about 8 years prior to my retirement. I have always had an interest in science subjects, particularly anything medical as both my parents and their siblings suffered health problems which may be hereditary.

- Luda Coutts
-
My name is Luda, I live in beautiful Stonehaven with my husband and collie dog. I am a pilates teacher and I am enjoying teaching my classes and see people moving and getting happy and healthy.

I feel privileged to be part of the PPI group, there is always a very friendly atmosphere, everyone's opinion is very welcome and I always looking forward to the next meeting.
- Caroline Keegan
-
My name is Caroline. I became interested in the research process when I was a Mental Health Nurse.

I feel very privileged to be involved with the work of the University of Aberdeen which has the potential to transform people's lives.
- Rossella Miccoli
-
Hi, my name is Rossella and I am an undergraduate student in French and German Studies at the University of Aberdeen. I joined the Patient & Public Involvement group in April as I believed it would be the perfect experience for an aspiring medical interpreter.

In 2021 I undertook an internship with a company in Germany specialised in medical support for foreigners in the country. There I was responsible for writing and translating articles for their blog and for hosting live interviews with their healthcare partners.
The PPI group is for me an invaluable resource to better understand our healthcare system and the progress of medical research. It allows me to get closer to my goal and keeps me encouraged and motivated.
- William Morrison
-
Retired for 20 years, I was employed in Local Government for just over 30 years finally as a Principal Officer in the Education department with responsibilities for all educational establishments.

My reasons for interest in the PPI group are mainly to keep the mind active and the possibility to contribute in a small way on the many and varied papers presented to us as a group.
- Gregory Poon
-
I spent my entire working life with NHS Grampian, my career consists of nursing, health promotion and management. Retired in 2010 after 41 years service.
I am a strong advocate in life science research as it lends important insights into disease processes and allows the

My experience with the PPI core group so far have been very positive. I learn a lot from the researchers and fellow PPI core group members. It is satisfying to hear feedback on how our contributions have made a difference.
- Ian Struthers
-
I became a member of the group as I have an interest in what research is being conducted in Aberdeen.
My background was engineering at university followed by a career in aviation.

I feel that as a lay person I can listen to briefings on research proposals then ask questions of, and provide feedback to, the presenters. People can sometimes be too close to a project and cannot see a relevant point which may be clearer to those who are less involved.
I enjoy the variety of research projects on which I have been briefed.
- David Wood
-
I come from a marine background and spent the majority of my career in the offshore environment. I have always been interested in voluntary work and before retirement was involved with Grampian Children's Panel and

Through Cafe Scientifique, I eventually became involved with the University of Aberdeen SMMSN PPI group. In parallel with this, through Cafe Med I became involved with the University of Aberdeen Patient Partner Programme.
Although there were some disruptions during the Covid lockdown, I continue to participate in both groups. I find the mental stimulation a welcome challenge, particularly as it is in complete contrast to my working background.
