Welcome to the North of Scotland Research Ethics Service (NoSRES).
NoSRES provides the administrative support for two Research Ethics Committees (North of Scotland Research Ethics Committee 1 - NoSREC 1 and North of Scotland Research Ethics Committee 2 - NoSREC 2). The service provides advice and support to all researchers at any time point in their careers.
NoSRES is part of a UK-wide national service aimed at facilitating research while protecting the rights, safety, dignity, and well-being of people participating in research within the NHS.
The remit of NoSRES mainly applies to research taking place within the NHS. If your research project does not come under our remit it can be reviewed via your Institutions Ethics Committee. For further advice don't hesitate to contact us.
All applications to NoSRES are made through the Integrated Research Application System (IRAS) https://www.myresearchproject.org.uk/. For further assistance/advice with IRAS please get in touch.
All projects that are submitted through IRAS require a Sponsor which is an organisation or individual responsible for the quality and conduct of a research project.
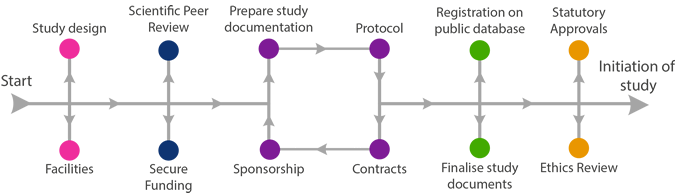
Unsure where to start your research journey? Below is a diagram which outlines the steps that you may need to undertake.