
BSc Hons, PhD
Lecturer
- About
-
- Email Address
- james.hislop@abdn.ac.uk
- Telephone Number
- +44 (0)1224 437399
- Office Address
6.19 Institute of Medical Sciences
- School/Department
- School of Medicine, Medical Sciences and Nutrition
Biography
I received my B.Sc in Pharmacology from the University of Bristol in 1997, before going on to undertake a Ph.D in the department of Medicine, also at the University of Bristol, under the supervision of Prof Craig McArdle, where I investigated the role of endocytosis in the function of the Gonadotropin Releasing Hormone Receptor. On completion of my PhD, I took a postdoctoral position at the University of California, San Francisco, where I continued my training in the laboratory of Prof Mark von Zastrow, a renowned leader in the field of endocytic trafficking in the function of G-protein-coupled receptors, where I investigated the role of Ubiquitin in endosomal trafficking. In 2011, I returned to the UK to work in the Gene Therapy Group at Imperial College London, in the laboratory of Prof Nicholas Mazarakis, where I studied how lentiviral vectors undergo trafficking within motor neurones. I was appointed Lecturer in Pharmacology at the University of Aberdeen in January 2013
Qualifications
- BSc Pharmacology1997 - University of Bristol
- PhD Pharmacology/Endocrinology2001 - University of Bristol
External Memberships
British Pharmacological Society
- Research
-
Research Overview
My research involves investigating the mechanisms by which G-protein-coupled Receptors are regulated to maintain signalling and homeostasis. G-protein coupled receptors (GPCRs), represent the largest family of cell surface receptors and are the prime regulators of many physiological systems. Their undoubted importance is demonstrated by the fact that as many as 50% of marketed drugs target GPCRs. Although there are >300 distinct GPCRs there are a number of similarities between how these different types are regulated, but also a number of differences, and it is these differences that have the most potential for therapeutic intervention.
The ability of a cell to respond to extracellular signals such as neurotransmitters is vital for regulating normal physiological function. One important aspect of this regulation is determining how many receptors are available to respond at any one time. Too few receptors and the signal is missed, too many and the signal overwhelms the cell. This process of desensitisation and resensitisation is important for the cell, and underlies, in part, the phenomenon of pharmaceutical tolerance. This signalling fidelity of GPCRs is maintained by an elegant series of interactions that firstly prevent signalling and is followed by a regulated trafficking process involving the removal of desensitised receptors from the surface of the cell (internalisation) followed by either recycling to the plasma membrane for repeated signalling (resensitisation) or targeting to the lysosome for destruction and downregulation (Figure 1). Although a great deal of progress has been made in understanding the processes underlying receptor endocytosis, including the critical role of receptor kinases and arrestin, comparatively little is known about the processes determining the postendocytic fate of GPCRs. My specific interests lie in how the levels of receptor are maintained or downregulated in the extended presence of agonist, and what mechanisms determine how receptors are sorted to the lysosome, in particular determining the distinct processes that control different receptor types.
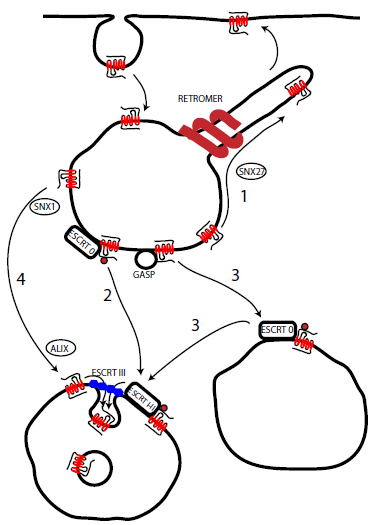
Figure 1 - Simplified model of GPCR post-endocytic sorting. Following endocytosis GPCRs can undergo recycling to the plasma membrane (1) (possibly regulated by the retromer) or sorting to the lysosome. This can involve ubiquitin regulated sorting by the ESCRT complex (2), initial ubiquitin-indendent sorting (possibly by GASP) before ubiquitin and ESCRT mediated transfer to Intralumenal vesicles (3) or direct interaction with the ESCRT III component, independent of ubiquitination (4) mediated by ALIX.
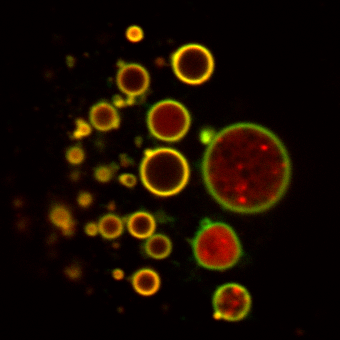
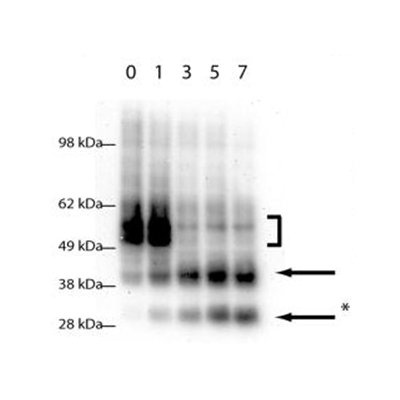
One area of particular interest is the role of ubiquitination in directing receptor translocation to intralumenal vesicles and its requirement or otherwise for trafficking to the lysosome. For many proteins ubiquitin is critical for controlling the degradation of receptors, but it is unknown what proteins are responsible for controlling this (e.g. the nature of the ubiquitin ligase) The images show a clear requirement for receptor (green) to be ubiquitinated or it remains on the endosome limiting membrane. However it is still able to undergo limited proteolysis with N-terminal fragments (red) being seen within the lumen of the endosome in some cases. This fragementation can be further visualised by Western blot (Henry et al, 2011).
Research Areas
Research Specialisms
- Pharmacology
- Neuroscience
- Cell Biology
Our research specialisms are based on the Higher Education Classification of Subjects (HECoS) which is HESA open data, published under the Creative Commons Attribution 4.0 International licence.
Current Research
Current Projects
- Investigating the link between endocytic trafficking and signalling
- The role of endocytic trafficking of FPR2 in the regulation of the inflammatory response (In collaboration with Dr D Thompson)
- The role of ubiquitination in the regulation of muscarinic acetylcholine receptors
- The role of downregulation of beta adrenergic receptors in cellular signalling
- Cannabinoid Receptor expression and function in the development of colorectal and prostate cancer (Funded by Friends of Anchor and NHS Grampian)
- Signalling Bias and trafficking and the development of novel compounds at cannabinoid receptors (In collaboration with Prof M Decker)
Post-Graduate Positions available
I am taking applications for self funded Masters students for any of the research areas above. Please contact me if you are interested.
Funding and Grants
Current and Previous Funders:
- BBSRC - EastBio-DTP
- Friends of Anchor
- NHS Grampian
- Tenovus Scotland
- Teaching
-
Teaching Responsibilities
I currently teach on and am the course coordinator of the Honours year undergraduates module Molecular Pharmacology (PA4005). I also lecture on the BM3501 - Cardiovascular Physiology and Pharmacology, and BM3502 - Neuroscience and Neuropharmacology and Molecular Pharmacology on the Clinical Pharmacology MSc course (MT5024). I supervise undergraduate and MSc students for lab based research projects
- Publications
-
Page 4 of 4 Results 31 to 38 of 38
Signaling and antiproliferative effects of type I and II gonadotropin-releasing hormone receptors in breast cancer cells
Journal of Clinical Endocrinology and Metabolism, vol. 89, no. 4, pp. 1823-32Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.1210/jc.2003-030787
Signalling and anti-proliferative effects mediated by gonadotrophin-releasing hormone receptors after expression in prostate cancer cells using recombinant adenovirus
Journal of Endocrinology, vol. 176, no. 2, pp. 275-84Contributions to Journals: ArticlesSignalling, cycling and desensitisation of gonadotrophin-releasing hormone receptors
Journal of Endocrinology, vol. 173, no. 1, pp. 1-11Contributions to Journals: ArticlesThe gonadotrophin-releasing hormone receptor: signalling, cycling and desensitisation
Archives of Physiology and Biochemistry, vol. 110, no. 1-2, pp. 113-122Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.1076/apab.110.1.113.893
Signaling and antiproliferative effects mediated by GnRH receptors after expression in breast cancer cells using recombinant adenovirus
Endocrinology, vol. 142, no. 11, pp. 4663-72Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.1210/endo.142.11.8503
Differential internalization of mammalian and non-mammalian gonadotropin-releasing hormone receptors. Uncoupling of dynamin-dependent internalization from mitogen-activated protein kinase signaling
The Journal of Biological Chemistry, vol. 276, no. 43, pp. 39685-94Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.1074/jbc.M104542200
Desensitization and internalization of human and xenopus gonadotropin-releasing hormone receptors expressed in alphaT4 pituitary cells using recombinant adenovirus
Endocrinology, vol. 141, no. 12, pp. 4564-75Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.1210/endo.141.12.7813
The lack of gonadotrophin-releasing hormone (GnRH) receptor desensitisation in alphaT3-1 cells is not due to GnRH receptor reserve or phosphatidylinositol 4,5-bis-phosphate pool size
Molecular and Cellular Endocrinology, vol. 147, no. 1-2, pp. 161-173Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.1016/S0303-7207(98)00201-9
