
PhD
Lecturer
- About
-
- Email Address
- j.holland@abdn.ac.uk
- Office Address
-
Lecturer in Fish Health and Aquaculture School of Biological Sciences
University of Aberdeen Room 2:14, Zoology Building
Tillydrone Avenue Aberdeen AB24 2TZ
TEL: +44 (0)1224 273228
FAX: +44 (0)1224 272396
- School/Department
- School of Biological Sciences
Biography
I am a Lecturer in Fish Health and Aquaculture
I received my PhD in Fish Immunology from the University College Swansea in 1998. I joined the Scottish Fish Immunology Research Centre at the University of Aberdeen in 1999 as a PDRA studying the molecular crosstalk between the immune and neuroendocrine systems in rainbow trout. I was appointed as senior research fellow in 2013 and currently a member of the oomycete research group at the Institute of Medical Sciences, University of Aberdeen. My interests are focused on the characterization of molecular mechanisms underpinning fish host-pathogen interactions with the view of developing novel immune therapies, particularly in the context of vaccine development. I have spearheaded proliferative kidney disease (PKD) research since 2007 through grants from the BBSRC, Swiss National Science Foundation, and the European Commission under the H2020 Programme (Grant H2020-634429 ParaFishControl) to develop genomic and functional tools to aid the shortlisting of parasite virulence factors and vaccine candidates. Currently, I am working towards the develop a novel immersion vaccination approach that has the potential to protect fish from a wide range of fish pathogens.
Qualifications
- BSc Biochemistry1993 - Swansea University
- PhD Fish Immunology1997 - Swansea University
External Memberships
Professional affiliations
I am a member of the:
European Association of Fish Pathologists
International Society of Comparative Immunology
British Society of Parasitology
International Veterinary vaccinology Network
BactiVac-acceleratiing bacterial vaccines
As part of the Aquaculture Research Collaborative Hub (ARCH) UK, I am the Finfish Health and Welfare working group lead
Peer Review
Journals:
Scientific Reports
Frontiers in Immunology
Trends in Parasitology
PLOS One
Journal of Experimental Biology
Fish and Shellfish Immunology
Developmental and Comparative Immunology
Molecular Immunology
Fish Biochemistry and Physiology
Veterinary Research
Diseases of Aquatic Organisms
Research funding agencies:
BBSRC
Austrian Science Fund
NSFC-ISF Joint Research Programme
Czech Science Foundation
BARD US-Israel Agricultural Research & Development Fund
- Research
-
Research Overview
RESEARCH
Research interests
1) Comparative Immunology
2) Vaccine development and immune therapies
3) Fish-parasite transcriptomics
4) Mechanisms of parasite virulence
Aquaculture is one of the fastest food production industries worldwide, with the salmon industry alone worth an estimated 800M to the Scottish economy per annum. Despite such growth, the future economic competitiveness and viability of the industry faces a number of challenges, particularly concerning losses due to fish disease. For successful disease control, a holistic understanding of fish immunity and mechanisms of pathogen virulence are important pre-requisites in developing new therapies.
My research focuses on the application of cell and molecular biology to characterise mechanisms that drive fish disease. Specifically, the detection and functional characterisation of fish immune and pathogen gene targets with the ultimate aim of developing novel vaccines and other therapeutic tools to combat fish diseases.
Pathogens which persist in or on fish hosts must, in some way, subvert or slow host immune responses to increase the likelihood of survival. In this context, I am increasingly interested in understanding the molecular mechanisms underpinning host-pathogen interactions, the functional implications of such interactions on immune responsiveness, and how this relates to pathogen control. Through the identification of immune pathways exploited by fish pathogens, the ability to develop immune therapies to replenish host immune responsiveness is likely. Elucidation of the pathogen molecules responsible for immune subversion will also aid development of effective vaccination strategies.
I have a long term interest in the control of proliferative kidney disease (PKD), a major parasitic disease affecting trout farming worldwide. Work in elucidating the underlying immune mechanisms driving PKD pathology has highlighted immune pathways and molecules that could be exploited therapeutically to eleviate chronic disease. Development of novel functional tools and parasite transcriptomes have greatly enhanced our understanding of the parasites biology, facilitated antigen selection for vaccine studies, and uncovered mechanisms by which myxozoan parasites may evade immunity.
Funded Research
2019-2020 (UKRI-BBSRC): “Passive and active immunization against novel vaccine targets to protect trout against proliferative kidney disease (PKD)”. DNA and protein-based vaccine studies following in silico selection and functional validation of lead vaccine candidates. Use of a TbMEG-1 SCAB construct to block antigen-host cell interactions by passive immunization.
2015-2019 (European Commision-funded PhD studentship): “ParaFishControl: Advanced Tools and Research Strategies for Parasite Control in European farmed fish”. My involvement centres around; the use of new and existing transcriptome assemblies to further facilitate vaccine development and to exploit immune molecules to eleviate disease pathology.
2014-2017 (SNSF): “Temperature driven emergence of Proliferative Kidney Disease (PKD) in salmonid fish”. My role is to transcriptomically and functionally characterize the parasite-vector interaction that drives PKD in salmonid fish.
2013-2015 (BBSRC): “Prophylactic measures in rainbow trout aquaculture: Further development of a DNA vaccine for PKD”. This project was instrumental in developing host-specific parasite transcriptomes to facilitate vaccine studies.
2007-2010 (BBSRC): “Towards the development of a vaccine for proliferative kidney disease”. This project utilised Expression Library Immunization and in silico antigen selection to take the first steps towards vaccine development.
Research Areas
Accepting PhDs
I am currently accepting PhDs in Biological and Environmental Sciences.
Please get in touch if you would like to discuss your research ideas further.

Research Specialisms
- Molecular Biology
- Parasitology
- Immunology
- Veterinary Pathology
- Transcriptomics
Our research specialisms are based on the Higher Education Classification of Subjects (HECoS) which is HESA open data, published under the Creative Commons Attribution 4.0 International licence.
Current Research
Research projects
Development of a novel approach to immersion vaccination
The control of fish disease remains one of the biggest challenges to the sustainability of fish aquaculture. Most existing vaccines are currently administered via injection, which is both costly and stressful to fish due to handling. Indeed, fish handling is one of the biggest causes of stress in aquaculture often leading to increased disease susceptibility in the immediate aftermath. Therefore, we are looking into developing an immersion based fish vaccination method, which would not require handling of the fish.
In a BBSRC/NERC funded project, we are now testing recombinant immersion vaccines on salmonid fish to determine whether they give protection to disease. Our work has the potential to revolutionise fish vaccination in the aquaculture sector.
Cell and molecular mechanisms underpinning proliferative kidney disease (PKD)
Proliferative kidney disease is a major parasitic disease affecting trout aquaculture with economic losses in the UK in excess of 2.5 million per annum. The causative agent, Tetracapsuloides bryosalmonae, belongs to a large enigmatic group of endoparasites, called the myxoxoans. From our discovery of phylogenetically restricted genes and phylogenomic studies by other researchers, we now know that these parasites are related to jellyfish.
Myxozoans begin their life cycles in aquatic invertebrates. In the case of T. bryosalmonae, it parasitises colonial invertebrates called bryozoans. The temperature driven release of spores from bryozoan colonies leads to the infection of farmed rainbow trout located downstream in river catchments. Parasite development in fish kidney tissue provokes an abnormal chronic proliferative response of host cells within the kidney giving rise to the gross kidney swelling that is so characteristic of the disease. Rainbow trout act as dead-end hosts, in that parasite development into renal stages capable of infecting naïve bryozoans when released in urine, does not occur. Compromised kidney function, stress, and increased susceptibility to other fish pathogens are the reasons for the considerable losses that can occur in naïve fish stocks.
There are no current vaccines or other therapeutic agents available to control PKD, although fish do develop protective immunity to re-infection, which provides the necessary impetus for vaccine development. Indeed farmers use this principle in pre-exposure programmes in an attempt to protect fish stocks, although production bottlenecks are inevitable.
My PKD research initially focussed on the underlying immune mechanisms that shape the kidney swelling response in infected fish. Kidney pathology is apparently refractory towards pro-inflammatory responses, although eliciting dominant anti-inflammatory-like activities, and dysregulated B and T lymphocyte responses. I have used suppressive subtractive hybridisation to provide further insights into host responses. This proved particularly successful in uncovering parasite genes from infected bryozoans and novel bryozoan immune-related genes. These early studies provided the impetus for my BBSRC-funded PKD vaccine studies using single parasite genes as candidate DNA vaccines in combination with expression library immunization (ELI) to facilitate the identification of protective antigens. Although highly successful, at this stage we only able to identify putative parasite antigens from infected bryozoans owing to the inability to purify parasites from infected fish. This has been a long-standing obstacle to PKD researchers owing to the very delicate nature of parasite spores in infected fish kidney tissue.
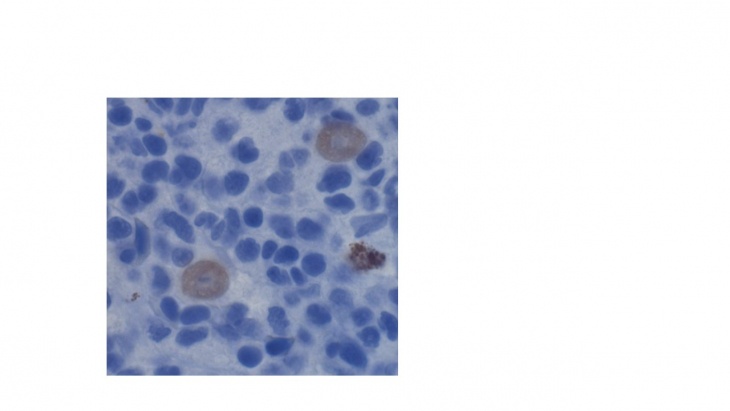
Figure 1. Extrasporogonic parasite stages within the interstitial kidney tissue of infected rainbow trout.
PKD transcriptomics and vaccine studies
To obtain parasite material from infected fish we have employed laser capture microdissection and, in a second BBSRC-funded project, transcriptomic approaches. We have succeeded in generating host-specific parasite transcriptome datasets. Parasite genes in common with both infected hosts have been annotated using a bioinformatic pipeline designed to enable the shortlisting of genes that are most likely to encode proteins of interest for vaccine development studies. Data concerning transcript coverage has been highly informative in allowing us to identify parasite genes that are likely to be fish-specific. Indeed, we have identified many genes confirmed to be fish-specific by real-time PCR analysis. This has greatly improved our knowledge of the parasites biology and mechanisms that may be operating to exacerbate disease pathogenesis, whilst providing an additional means of shortlisting fish-specific parasite antigens and putative virulence factors for functional and vaccine development studies. These studies are currently ongoing as part of the EU Horizon 2020-funded consortium project, ParaFishControl.
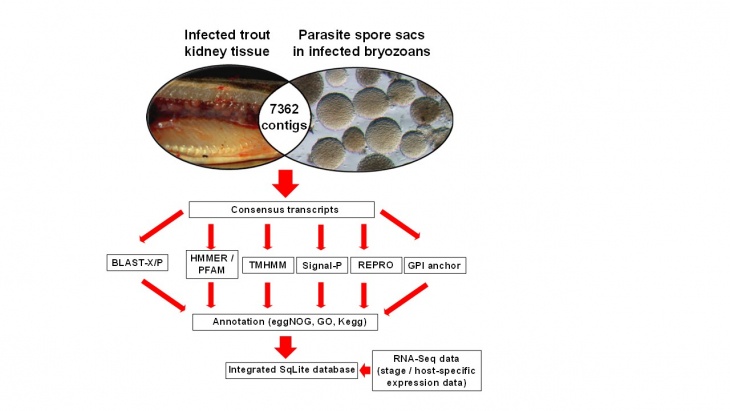
Figure 2. Bioinformatics pipeline used to shortlist parasite genes for vaccine studies based on parasite transcripts in common with both infected hosts (images courtesy of Marc Faber and Hanna Hartikainen).
Characterisation of bryozoan-parasite interactions
I recently completed a sub-project as part of a Swiss National Science Foundation (SNSF)-funded Sinergia consortium project. PKD is particularly problematic in brown trout populations is Swiss river catchments. To facilitate the identification of host and parasite biomarkers of infection that may inform on disease risk and spread in river systems, my main role was to transcriptomically and functionally characterize the parasite-invertebrate vector interaction that drives PKD in salmonid fish. Here, I have been able to identify many bryozoan-specific parasite genes and immune-related bryozoan molecules modulated by parasite infection. These observations have greatly increased our knowledge of the invertebrate host-parasite interaction and have highlighted a common link in the nature of immune responses in both infected hosts. Functional studies on the fish host have successfully identified a putative mechanism by which parasites are able to suppress pro-inflammatory responses in fish.
Collaborations
Collaborations
Chris Secombes, University of Aberdeen
Tiehui Wang, University of Aberdeen
Marc Faber, University of Aberdeen
Carolina Tafalla, CISA-INIA, Madrid, Spain
Aitor Granja, CISA-INIA, Madrid, Spain
Beth Okamura, Natural History Museum, London
Hanna Hartikainen, Eawag Aquatic Research, DÈ•bendorf, Switzerland
Thomas Wahli, University of Bern, Switzerland
- Teaching
-
Teaching Responsibilities
Current teaching responsibilities:
1st Year: BI1009: Frontiers in Biological Sciences (lectures & practicals)
3rd Year: Z03513: Environmental Physiology (lecture)
4th and 5th Year: ZO4811 / ZO5807: Sustainable Aquaculture (course coordinator; lectures)
4th Year: BI4026: Meet the Scientist: (course coordinator; lectures / tutorials)
Non-course Teaching Responsibilities
9-month PhD student progress assessment examiner
