
BSc (Durham), PhD (Cambridge)
Personal Chair
- About
-
- Email Address
- a.c.mclaughlin@abdn.ac.uk
- Telephone Number
- +44 (0)1224 272924
- Office Address
Meston Room G23
- School/Department
- School of Natural and Computing Sciences
Biography
Professor Abbie Mclaughlin is currently a chair in inorganic chemistry at the University of Aberdeen. Her expertise lies in the synthesis and study of transition metal oxides with fascinating electrical and magnetic properties. This includes discovery of novel exotic magnetic and electronic materials and novel electrolytes for application in ceramic fuel cells. She is particularly interested in structure-property relationships and determining design rules to improve a specific property.
She has published ~ 80 papers (h-index 23, Google Scholar) on the study of transition metal oxides using structural (powder diffraction) and physical (magnetic and conductivity) measurements. She has secured over £2.5M research funding from sources such as EPSRC, the Leverhulme Trust, the Royal Society of Edinburgh and the Royal Society. She has been awarded > 200 days of neutron diffraction and synchrotron X-ray diffraction beamtime at the large-scale facilities of ISIS and Diamond, Didcot and the ILL and ESRF, Grenoble.
She is a member of the EPSRC college and serves on the EPSRC Physical Sciences panel. She has previously served on ISIS, ILL and Oakridge facility access panels.
Memberships and Affiliations
- Internal Memberships
-
Professor Mclaughlin is currently Head of the Department of Chemistry.
- External Memberships
-
FRSC
Prizes and Awards
2021 University of Aberdeen Excellence Award - Outstanding Research Awards: Individuals Further Stage of Career – STEM.
2020-2021 Leverhulme Trust research fellowship.
2006-2009 Leverhulme Trust early career fellowship.
2003-2006 Royal Society of Edinburgh, SEELLD personal research fellowship.
- Research
-
Research Overview
Solid state materials are of fundamental importance due to the plethora of exciting physical properties that have been detected and applied in technological applications. We are interested in synthesising novel materials that exhibit fascinating electrical (ionic conducting) or magnetic properties.
Rietveld refinement of crystallographic models from X-ray and Neutron powder diffraction data (at facilities such as ISIS, Rutherford Appleton Laboratory, Didcot, UK and the ESRF and ILL, Grenoble, France) are used in order to correlate the structure-property relationships in the novel synthesised compounds which enable us to tune the properties of our materials.
Research Expertise
Solid state synthesis
Rietveld refinement of crystallographic models from X-ray and neutron powder diffraction data
Characterisation of ionic conductors via impedance spectroscopy
Characterisation of superinsulators
Electronic and magnetic characterisation of novel oxides and pnictides
Magnetic structure determination
Research Facilities
A suite of high temperature tube and box furnaces (up to 1600 oC) for solid state synthesis
Two top of the range diffractometers (Panalytical X’Pert Powder and Empyrean powder diffractometers)
Variable temperature impedance jig coupled to a Solartron SI 1260 Impedance/Gain-Phase Analyser
Stanton Redcroft TG/DTA
Departmental access to a Mettler Toledo TGA 2 coupled with a Hiden Quadrupole Mass Spectrometer
Departmental access to a Carl Zeiss Gemini SEM 300 to determine the microstructures of samples in addition to EDX measurements
Research Areas
Accepting PhDs
I am currently accepting PhDs in Chemistry.
Please get in touch if you would like to discuss your research ideas further.
Research Specialisms
- Inorganic Chemistry
- Materials Science
- Condensed Matter Physics
- Chemistry
Our research specialisms are based on the Higher Education Classification of Subjects (HECoS) which is HESA open data, published under the Creative Commons Attribution 4.0 International licence.
Current Research
Oxide Ion Conductors
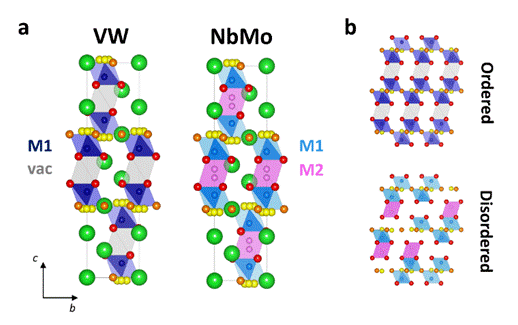
Solid oxide fuel cells (SOFCs) are electrochemical devices able to generate electricity from renewable energy sources, with high efficiency and low emissions of pollutants. However, oxide ion transport in most commercially available solid electrolytes is enabled only at high working temperatures (> 700 °C), thus making the commercialization of SOFCs particularly challenging. The high operating temperature reduces the components’ durability, results in slow start up times and the necessitation of expensive materials for seals and interconnects. In order to overcome these issues, it is desirable to develop novel electrolyte materials with significant oxide ion conductivity at intermediate temperatures (300 – 600 °C). Ionic conduction in solid oxides is strongly related to the crystal structure, and as a result the discovery and characterization of novel oxide ion conductors in various structural families has attained significant attention from researchers. We have been investigating hexagonal perovskites Ba3M’M’’O8.5 (M’M’’ = VW, NbMo, NbW) which exhibit highly disordered crystal structures and state-of-the art conductivity. We are currently interested in synthesising new materials with different combinations of M’M’’ to see how the chemistry controls the ionic conductivity.
Mixed Proton/Oxide Ion Conductors
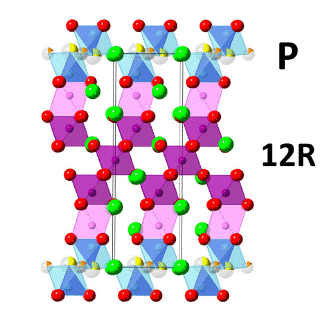
We are also interested in mixed ion conductors, specifically mixed oxide-ion and proton-conductors. Such dual ion conductors have been proposed as a new class of electrolyte for intermediate temperature ceramic fuel cells, as they exhibit low ohmic resistance without external gas humidification. We have been investigating the electrical and structural properties of Ba7Nb4MoO20, a cation-deficient 7H hexagonal perovskite derivative formed by an intergrowth of palmierite layers and 12R perovskite blocks. Ba7Nb4MoO20 supports remarkable oxide ion and proton conductivity with excellent chemical and electrical stability over a range of pO2. The ionic conduction properties of Ba7Nb4MoO20 are linked to its distinct disordered crystal structure. Ba7Nb4MoO20 supports pure ionic conduction with high proton and oxide ion conductivity at 510 °C (the bulk conductivity is 4.0 mS cm-1) and hence is an exceptional candidate for application as a dual-ion solid electrolyte in a ceramic fuel cell which will combine the advantages of both oxide ion and proton conducting electrolytes. We are currently performing chemical doping studies to further enhance the conductivity.
Magnetic Materials
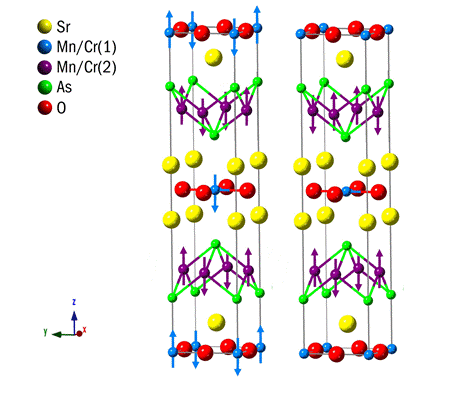
The recent report of high temperature superconductivity in oxypnictides such as LnFeAsO1-xFx with superconducting transition temperatures (Tcs) up to 55 K has led to a rapid expansion in the research of oxypnictide materials. We have recently discovered the counterpart, colossal magnetoresistance (CMR) in Mn2+ based oxypnictides NdMnAsO1-xFx, so that both manifestations of correlated electronic behaviour are now found in these new materials. Colossal magnetoresistant materials exhibit a large change (> 90%) in their electronic resistivity upon application of a magnetic field and have application as magnetoresistive sensors and spintronic devices in which electron spins are used to process information. We are currently investigating more complex cation ordered oxypnictides such as Sr2Mn2CrAs2O2 which exhibit spin reorientation magnetic transitions upon cooling which may show coupling between magnetic and electronic degrees of freedom.
Funding and Grants
We have had funding from a range of sources including EPSRC, the Leverhulme Trust, Institut Laue Langevin, the Commonwealth Scholarship Commission, Chief Scientist Office and the Royal Society.
If you’re interested in joining the group as a PhD student, please email a.c.mclaughlin@abdn.ac.uk.
- Teaching
-
Teaching Responsibilities
Dr Mclaughlin teaches the following courses:
- CM1022 Elements of Chemistry
- CM2519 Solids
- CM2012 Electrical Materials
- CM3037 Transition Metal Chemistry
- CM4026 Lanthanides and actidnides
- CM5003 Magnets, metals and superconductors
- Publications
-
Page 1 of 9 Results 1 to 10 of 85
Structure Property Relationships of the Exotic Insulator-Insulator Transition in CeMnAsO1-xFx: A Potential Excitonic Insulator
Chemistry of MaterialsContributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.1021/acs.chemmater.6c00090
The Structure and Electrical Properties of the Palmierite Sr3P2O8
Journal of Solid State Chemistry, 125962Contributions to Journals: ArticlesElucidating oxide-ion and proton transport in ionic conductors using machine learning potentials
npj Computational Materials, vol. 11, no. 1, 328Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.1038/s41524-025-01807-y
- [OPEN ACCESS] http://aura.abdn.ac.uk/bitstreams/c0591a4d-6d92-47af-a17e-7b6c415d0830/download
- [ONLINE] View publication in Scopus
Enhancement of oxide ion and proton conductivity in Sr3-xCaxV2O8 palmierites through tuning of the crystal structure
Solid State Ionics, vol. 432, 117041Contributions to Journals: ArticlesEnhanced Oxide Ion Diffusion by Lanthanum Substitution in the Palmierite Sr3-3xLa2xV2O8 via Increased Tetrahedral Distortion and Cation Vacancies
Chemistry of Materials, vol. 37, no. 19, pp. 7987-7995Contributions to Journals: ArticlesUnlocking the potential of palmierite oxides: high oxide ion conductivity via induced interstitial defects
Journal of the American Chemical Society, vol. 147, no. 11, pp. 9694-9703Contributions to Journals: ArticlesTuning of the Ionic Conductivity of Ba7Nb4MoO20 by Pressure: A Neutron Diffraction and Atomistic Modelling Study
Journal of Materials Chemistry A, vol. 13, no. 6, pp. 4444-4451Contributions to Journals: ArticlesDevelopment of Nanostructured Lanthanum Strontium Cobalt Ferrite/Gadolinian-Doped Ceria Composite Electrodes of Solid Oxide Cells Formed by In Situ Polarization
ACS Applied Materials & Interfaces, vol. 16, no. 17, pp. 21818–21827Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.1021/acsami.4c01223
- [OPEN ACCESS] http://aura.abdn.ac.uk/bitstreams/bc9cc35c-c702-439c-bce0-fbda5590952f/download
- [ONLINE] View publication in Scopus
A Variable Temperature Neutron Diffraction Study of Dual Ion Conducting Sr3V2O8
Journal of Solid State Chemistry, vol. 331, 124512Contributions to Journals: ArticlesObservation of an Exotic Insulator to Insulator Transition upon Electron Doping the Mott Insulator CeMnAsO
Nature Communications, vol. 14, 1737Contributions to Journals: Articles

