By studying the associated in situ biota in the individual chert beds, much information can be gathered on the interactions between fauna, flora and the inorganic environment at Rhynie during the Early Devonian some 400 million years ago, thus helping to build a picture of this early terrestrial and freshwater ecosystem.
Of course, to understand how an ancient palaeoecosystem worked, such as the food chain, trophic resources, habitat space, etc. is extremely difficult for three main reasons. Fossils cannot be observed as living organisms; many environmental parameters, including modes of life, are not necessarily directly comparable with modern examples; and the fossil record is strongly influenced by taphonomy, biostratinomy and diagenesis and thus only organisms capable of being fossilised are generally preserved.
However, the remarkable preservation of the biota in the Rhynie chert means that although by no means complete, the palaeoecology of this deposit can be better understood than that of many other fossil assemblages. There are three types of 'interaction' in an ecosystem:
- Coaction: where an organism affects another organism.
- Reaction: where an organism affects the inorganic environment.
- Action: where the inorganic environment affects an organism.
Evidence, to a greater or lesser degree, has been found of all three interaction types in the different beds of chert.
- Evidence for Coaction
-
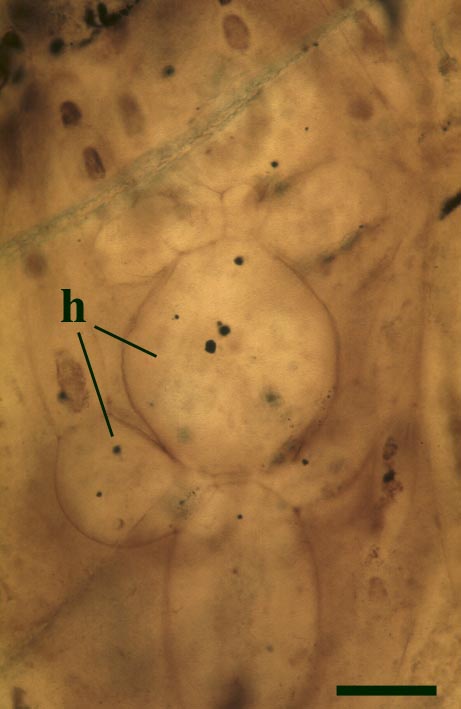
Most living organisms survive by directly or indirectly exploiting others, such organisms are incapable of producing food from inorganic substances, with the exception of green plants and algae which produce carbohydrate from photosynthesis. Few organisms, even green plants, live entirely independently, nearly all survive as parts of a living community. For example, certain organisms live by predation, others by parasitism, other organisms may be herbivorous, and others saprophytic, living off dead organic matter. Perhaps the greatest degree of interdependence occurs when two different organisms live attached to one another in an association which is mutually beneficial to both organisms, known as symbiosis.

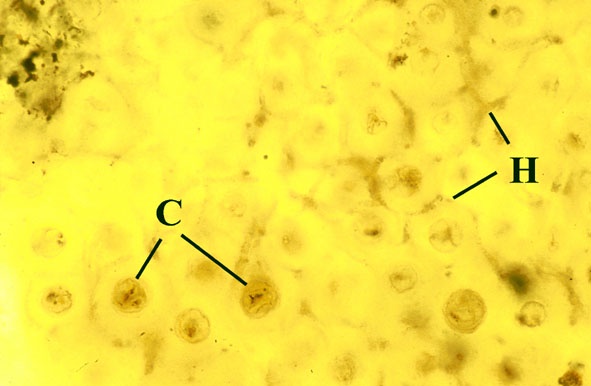
It is clear from understanding mycorrhizae and lichens that symbiosis is an extremely widespread phenomenon, essential to the normal functioning of nearly all modern plant communities and was clearly developed in Early Devonian terrestrial communities.
The most likely predators found in the Rhynie chert are the trigonotarbid arachnids and centipedes, since both display morphological characteristics that lend them to a predatory lifestyle, namely the possession of fangs and poison claws respectively; though no unequivocal evidence has been found to date to identify specific prey items. An interesting feature with the trigonotarbids however, is their common association with empty sporangia and plant straws ( Kevan et al. 1975 ). These may have provided the arachnid with a hiding place from which to capture prey or alternatively may have provided a relatively safe shelter in which the animal could moult its cuticle. Some authors have suggested the Rhynie trigonotarbids may have been facultative herbivores, occasionally feeding on spore protoplasts ( Kevan et al. 1975 ).
A number of the Rhynie organisms appear to have been wholly or at least partially saprophytic. As mentioned above, the mites and the collembolan were likely to have been partially saprophytic in their ecology. At least some of the Rhynie fungi were also likely to be saprophytic, evidenced by the close association of fungal hyphae with decayed plant material. But perhaps the most unequivocal evidence of this form of coaction, and detritivory, is the preservation of the gut contents in the myriapod Leverhulmia mariae, found to comprise almost entirely spores and macerated plant debris ( Anderson & Trewin 2003 ). The euthycarcinoid Heterocrania rhyniensis and the crustacean Lepidocaris rhyniensis were likely to be saprophytes and detritivores as evidenced by their morphology, particularly in their mouthparts, and more speculatively from the contents of coprolites, found in close association, that may have been produced by these organisms.
- Evidence for Reaction
-
The evidence for reaction in the Rhynie chert (i.e.: the evidence for the biota affecting the inorganic environment) is less clear than that for evidence of coaction, and perhaps more speculative. Perhaps the best evidence for reaction is the contribution of the biota, particularly the flora to soil formation.

A number of the cyanobacteria found in the Rhynie chert, including Archaeothrix possess heterocysts. Heterocysts are used by modern photosynthetic bacteria in the fixation of atmospheric nitrogen. It is very likely, therefore that many of the Early Devonian cyanobacteria played an important roll in fixing atmospheric nitrogen into soils at Rhynie 400 million years ago.
- Evidence for Action
-
Evidence for action in the Rhynie chert is rather subjective, particularly since we cannot see the Rhynie biota as living organisms. It would be expected, considering the presence of an active hydrothermal complex at Rhynie 400 million years ago, that the 'inorganic environment' would have had some affect on the biota. However, we know that a number of different 'sub-environments' existed within this complex.
For example, we have seen in certain chert beds containing a rich biota of crustaceans, euthycarcinoids, charophytes and various forms of chytrid fungi, algae and cyanobacteria, that these particular beds were deposited in localised pools and ephemeral bodies of standing water. It is not likely that these pond-dwelling organisms were particularly specialised for a hot-water environment. With the exception of cyanobacteria, most 'modern equivalents' of these organisms would not tolerate high water temperatures. It is possible, therefore, that these 'aquatic assemblages' may have represented a background biota that in life was not adapted to life in hot springs, but were killed and preserved by the sudden invasion of their living environment by hot spring fluids.
However, it has been noted in modern hydrothermal areas that in certain areas in proximity to hot-springs and geysers that the flora may be specialised and some forms stunted, reflecting variations in available water, water temperature, pH and nutrients. It remains a matter of debate as to whether some of the Rhynie flora were also specialised in this way or if they represented the background flora living at the time. Current studies on spore assemblages from the cherts and interbedded sediments may help to answer this question.
