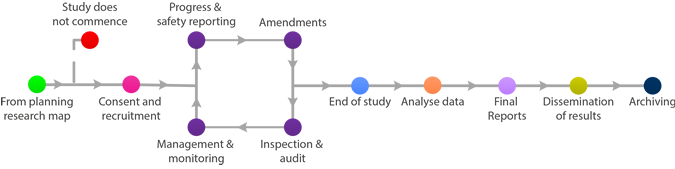
Other important considerations relating to statistical analysis include ensuring that, wherever possible, the trial statistician remains blinded until data analysis is complete.
There must be a quality check of data prior to interim and final analyses. Quality checks should include checking for outliers, missing data, date checks and any inconsistencies. In some cases, the Sponsor may impose stringent criteria on who can analyse trial data and care must be taken to ensure no bias is brought into the results.
When analysis is complete, the trial results should be discussed by the Chief Investigator and all relevant oversight groups (such as Data Monitoring Committee and Trial Steering Committee) to assist interpretation and to discuss the implications of the findings.
Visit the SOPs and Templates page for further guidance.